What is a Tunnel Effect Microscope?
A tunnel microscope (STM) is a device for capturing images of atomic surfaces. The tunneling microscope allows the use practices of the current of electrons that originates between a very fine preceptor tip and a conductive prototype, when they are placed very close and a potential is used between both. The image that is collected is the electronic density on the conductive surface allowing atomic order resolutions. The sensitivity of this type of microscope is very high because the current of electrons decreases exponentially with the separation or increase of distance between the tip and the surface to be observed.
What is a Confocal Microscope?

Within the laboratory, the use of microscopes is necessary for conducting biological research. Basically, it is the use of light sources, light detectors and other elements that prevent luminescence from planes other than the focus of the sample. Its use has spread in different fields of science, specifically in biomedical.
What are the steps to use and calibrate a meter?

It is important to follow the instructions and recommendations of manufacturers for the use and calibration of a phmeter, since, to achieve a correct measurement with a phmeter must be calibrated correctly, remember that the pH measures the concentration of hydrogen ions, which determine the degree of acidity and basicity of a solution; in addition not only works to measure the degree of water contamination, but as a diagnosis of body diseases or when measuring pH of urine and blood.
PH meter, water pH, milk pH, and human body pH

PH is a parameter used to measure the concentration of the hydrogen ion [H +] in a solution. In definition pH, it is the negative logarithm of base 10 of the activity of the [H +]. PH values vary on a scale from 0 to 14, which indicates the degree of acidity or alkalinity of an aqueous solution.
How do I use a laboratory pH meter?

A pH meter or pH meter is a laboratory instrument used to measure the acidity or alkalinity of a solution, perform a fairly accurate analysis on these fluids, this is done through polarizable glass plates sensitive to hydrogen ions.
Importance of pHmeter in a laboratory

A pH meter is one of the most used equipment in a laboratory. It is used to measure the acidity or alkalinity of a solution and the information it provides expresses the degree of acidity of an acid or base in terms of the activity of hydrogen ions, so through the use of this equipment we can know the type of substance we are analyzing, as well as the type of treatment that should be given to it
PH meter or pH meter: How should it be used?

A pH meter is an instrument used to measure the acidity or alkalinity of a solution. The information provided by this equipment expresses the degree of acidity of an acid or base in terms of the activity of hydrogen ions.
What is pH and how is it measured?

pH is the unit of measurement that describes the degree of acidity or alkalinity and is measured on a scale that goes from 0 to 14. The measurement of pH (hydrogen potential) is one of the most important parameters in a laboratory, it has a great field of application such as: research, industry, teaching, among others. In laboratories where it is relevant to know the potential of hydrogen, a pH measurement equipment will always be found as a primary instrument.
Laboratory pH meter, working

A pH meter or pH meter is a laboratory equipment used to measure the acidity or alkalinity of a solution, that is, it is an instrument whose function is to measure the pH level of liquids through glass plates, and thus way to accurately obtain an analysis on these fluids. The information provided by this laboratory device expresses the degree of acidity of an acid or a base in terms of the activity of hydrogen ions.
What is a pH meter? What is pH?
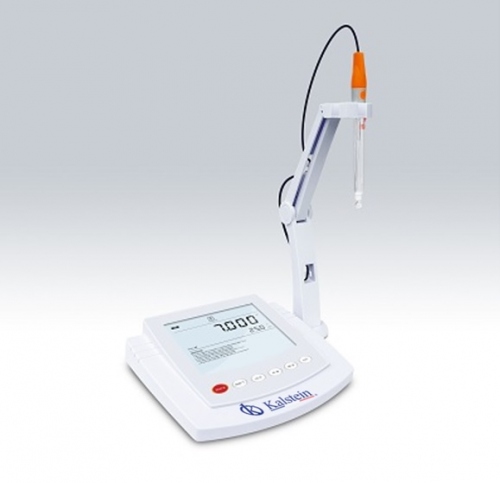
A pH meter is a laboratory equipment that is used to accurately measure the degree of acidity or alkalinity of a solution. This equipment provides information that expresses the degree of acidity of an acid or a base in terms of the activity of hydrogen ions.
